Pergoveris 900 / 450iu Pen Follitropin alfa Lutropin alfa, 1 pack
Multidose Prefilled Pen
Treatment of Infertility in Women
Active Ingredient: Follitropin alfa and Lutropin alfa
Prescription Required
- Multidose Prefilled Pen
- Treatment of Infertility in Women
- Active Ingredient: Follitropin alfa and Lutropin alfa
Original price was: £774.00.£630.15Current price is: £630.15.
CompareCompare- Multidose Prefilled Pen
- Treatment of Infertility in Women
- Active Ingredient: Follitropin alfa and Lutropin alfa
Pergoveris 900 / 450iu Pen Follitropin alfa Lutropin alfa, 1 pack
What is Pergoveris 900?
Pergoveris 900 contains two different active substances called “follitropin alfa” and “lutropin alfa”. Both
belong to the family of hormones called “gonadotropins”, which are involved in reproduction and
fertility.
What Pergoveris 900 is used for?
Pergoveris 900 is used to stimulate the development of follicles (each containing an egg) in your ovaries. This is to help you get pregnant. It is for use in adult women (18 years old or above) who have low levels (severe deficiency) of “follicle stimulating hormone” (FSH) and “luteinising hormone” (LH). These women are usually infertile.
How does Pergoveris work?
The active substances in Pergoveris are copies of the natural hormones FSH and LH. In your body:
• FSH stimulates the production of eggs
• LH stimulates the release of the eggs.
By replacing the missing hormones, Pergoveris allows women with low levels of FSH and LH to develop a follicle. This will then release an egg, after an injection of the hormone “human chorionic gonadotropin (hCG)”. This helps the women to become pregnant.
What is Infertility
Infertility is when a couple cannot get pregnant (conceive) despite having regular unprotected sex.
Around 1 in 7 couples may have difficulty conceiving.
About 84% of couples will conceive naturally within a year if they have regular unprotected sex (every 2 or 3 days).
For couples who have been trying to conceive for more than 3 years without success, the likelihood of getting pregnant naturally within the next year is 1 in 4, or less.
Some people get pregnant quickly, but for others it can take longer. It’s a good idea to see a GP if you have not conceived after a year of trying.
Women aged 36 and over, and anyone who’s already aware they may have fertility problems, should see their GP sooner.
They can check for common causes of fertility problems and suggest treatments that could help.
Infertility is usually only diagnosed when a couple have not managed to conceive after a year of trying.
There are 2 types of infertility:
- primary infertility – where someone who’s never conceived a child in the past has difficulty conceiving
- secondary infertility – where someone has had 1 or more pregnancies in the past, but is having difficulty conceiving again
Fertility treatments include:
- medical treatment for lack of regular ovulation
- surgical procedures such as treatment for endometriosis, repair of the fallopian tubes, or removal of scarring (adhesions) within the womb or abdominal cavity
- assisted conception such as intrauterine insemination (IUI) or IVF
The treatment offered will depend on what’s causing the fertility problems and what’s available from your local clinical commissioning group (CCG).
Private treatment is also available, but it can be expensive and there’s no guarantee it will be successful.
It’s important to choose a private clinic carefully. You can ask a GP for advice, and should make sure you choose a clinic that’s licensed by the Human Fertilisation and Embryology Authority (HFEA).
Some treatments for infertility, such as IVF, can cause complications.
For example:
- multiple pregnancy – if more than 1 embryo is placed in the womb as part of IVF treatment there’s an increased chance of having twins; this may not seem like a bad thing, but it significantly increases the risk of complications for you and your babies
- ectopic pregnancy – the risk of having an ectopic pregnancy is slightly increased if you have IVF
Read more about how infertility is treated.
There are many possible causes of infertility, and fertility problems can affect either partner. But in a quarter of cases it is not possible to identify the cause.
Common causes of infertility include:
- lack of regular ovulation (the monthly release of an egg)
- poor quality semen
- blocked or damaged fallopian tubes
- endometriosis – where tissue that behaves like the lining of the womb (the endometrium) is found outside the womb
RISK FACTORS
There are also several factors that can affect fertility.
These include:
- age – fertility declines with age
- weight – being overweight or obese (having a BMI of 30 or over) reduces fertility; in women, being overweight or severely underweight can affect ovulation
- sexually transmitted infections (STIs) – several STIs, including chlamydia, can affect fertility
- smoking – can affect fertility: smoking (including passive smoking) affects your chance of conceiving and can reduce semen quality; read more about quitting smoking
- alcohol – the safest approach is not to drink alcohol at all to keep risks to your baby to a minimum. Drinking too much alcohol can also affect the quality of sperm (the chief medical officers for the UK recommend adults should drink no more than 14 units of alcohol a week, which should be spread evenly over 3 days or more)
- environmental factors – exposure to certain pesticides, solvents and metals has been shown to affect fertility, particularly in men
- stress – can affect your relationship with your partner and cause a loss of sex drive; in severe cases, stress may also affect ovulation and sperm production
There’s no evidence to suggest caffeinated drinks, such as tea, coffee and colas, are associated with fertility problems.
Pergoveris Pen 300/150iu Reviews
After using Pergoveris Pen 300/150iu, it’s helpful to let others know about your experience. Reviews of an item help other users know that medicines received have helped the condition it is claimed for, how well the treatment worked or any issues to be aware of. We invite our users to leave a review of both their treatment and of the service provided. Click on the reviews tab to see if there has been feedback on this item.
Price of Pergoveris Pen 900/450iu in UK
Where to buy Pergoveris Pen 900/450iu
Pergoveris Pen 900/450iu is available to buy with a prescription at Dock Pharmacy Essex UK, UK Online Pharmacy.
Brand
Pergoveris
How To Use
How to use Pergoveris 900
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor
or pharmacist if you are not sure.
Using this medicine
• Pergoveris is intended to be given by injection just under the skin (subcutaneously). To minimise skin irritation, select a different injection site each day.
• Your doctor or nurse will show you how to use the Pergoveris pre-filled pen to inject the medicine.
• If they are satisfied that you can administer Pergoveris safely, you can then prepare and inject the medicine yourself at home.
• If you administer Pergoveris to yourself, please carefully read and follow section “Instructions for Use”.
How much to use
A treatment regimen commences with the recommended dose of Pergoveris containing 150 International Units (IU) of follitropin alfa and 75 IU of lutropin alfa every day.
• According to your response, your doctor may decide to add every day a dose of a licensed follitropin alfa preparation to your Pergoveris injection. In this case, the follitropin alfa dose is
usually increased every 7 or every 14 days by 37.5 to 75 IU.
• Treatment is continued until you get the desired response. This is when you have developed a suitable follicle, as assessed using ultrasound scans and blood tests.
• This may take up to 5 weeks.
When you get the desired response, you will be given a single injection of human chorionic gonadotropin (hCG) 24 to 48 hours after your last Pergoveris injection. The best time to have sex is on
the day of the hCG injection and the day after. Alternatively, intrauterine insemination (IUI) may be performed.
If your body responds too strongly, your treatment will be stopped and you will not be given any hCG (see in section 2. under ”Ovarian hyperstimulation syndrome (OHSS)”). In this case, your doctor will
give you a lower follitropin alfa dose in the following cycle.
If you use more Pergoveris than you should
The effects of an overdose of Pergoveris are unknown, nevertheless one could expect OHSS to occur.
However this will only occur if hCG is administered (see in section 2. under “Ovarian hyperstimulation syndrome (OHSS)”).
If you forget to use Pergoveris
Do not use a double dose to make up for a forgotten dose. Please contact your doctor.
If you have any further question on the use of this medicine, ask your doctor, pharmacist or nurse
Product Details
What you need to know before you use Pergoveris
You and your partner’s fertility should be evaluated before the treatment is started by a doctor
experienced in treating fertility problems.
Do not use Pergoveris
• if you are allergic to follicle stimulating hormone (FSH), luteinising hormone (LH) or any of the
other ingredients of this medicine (listed in section 6)
• if you have a brain tumour (in your hypothalamus or pituitary gland)
• if you have large ovaries or sacs of fluid within your ovaries (ovarian cysts) of unknown origin
2
• if you have unexplained vaginal bleeding
• if you have cancer in your ovaries, womb or breasts
• if you have a condition that would make a normal pregnancy impossible, such as an early
menopause, malformed sex organs or benign tumours of the womb.
Do not use this medicine if any of the above apply to you. If you are not sure, talk to your doctor,
pharmacist or nurse before using this medicine.
Warnings and precautions
Talk to your doctor, pharmacist or nurse before using Pergoveris.
Porphyria
Talk to your doctor before you start your treatment.If you or any member of your family have
porphyria (an inability to breakdown porphyrins that may be passed on from parents to children).
Tell your doctor straight away if:
• your skin becomes fragile and easily blistered, especially skin that has been frequently exposed
to sunlight
• you have stomach, arm or leg pain.
In case of above events your doctor may recommend that you stop treatment.
Ovarian hyperstimulation syndrome (OHSS)
This medicine stimulates your ovaries. This increases your risk of developing ovarian
hyperstimulation syndrome (OHSS). This is when your follicles develop too much and become large
cysts. If you get lower abdominal pain, gain any weight rapidly, feel sick or are vomiting or if you
have difficulty in breathing, talk to your doctor straight away. They might ask you to stop using this
medicine (see in section 4. under “Most serious side effects”).
In case you are not ovulating and if the recommended dose and schedule of administration are adhered
to, the occurrence of severe OHSS is less likely. Pergoveris treatment seldom causes severe OHSS.
This becomes more likely if the medicine that is used for final follicular maturation (containing human
chorionic gonadotrophin, hCG) is administered (see in section 3. under “How much to use” for
details). If you are developing OHSS your doctor may not give you any hCG in this treatment cycle
and you may be told not to have sex or that you should use a barrier contraceptive method for at least
four days.
Your doctor will ensure, careful monitoring of the ovarian response, based on ultrasound and blood
tests (oestradiol measurements) before and during the course of treatment.
Multiple pregnancy
When using Pergoveris, you have a higher risk of being pregnant with more than one child at the same
time (“multiple pregnancy”, mostly twins), than if you conceived naturally. Multiple pregnancy may
lead to medical complications for you and your babies. You can reduce the risk of multiple pregnancy
by using the right dose of Pergoveris at the right times.
To minimise the risk of multiple pregnancy, ultrasound scans as well as blood tests are recommended.
Miscarriage
When undergoing stimulation of your ovaries to produce eggs, you are more likely to have a
miscarriage than the average woman.
Ectopic pregnancy
Women who have ever had blocked or damaged fallopian tubes (tubal disease) are at risk of pregnancy
where the embryo is implanted outside the womb (ectopic pregnancy). This is whether the pregnancy
is obtained by spontaneous conception or with fertility treatments.
3
Blood clotting problems (thromboembolic events)
Talk to your doctor before using Pergoveris if you or a member of your family have ever had blood
clots in the leg or in the lung, or a heart attack or stroke. You may be at a higher risk of serious blood
clots or existing clots might become worse with Pergoveris treatment.
Tumours of sex organs
There have been reports of tumours in the ovaries and other sex organs, both benign and malignant, in
women who have undergone multiple regimens for infertility treatment.
Allergic reactions
There have been isolated reports of non-serious allergic reactions to Pergoveris. If you have ever had
this type of reaction to a similar medicine, talk to your doctor before using Pergoveris.
Children and adolescents
Pergoveris is not for use in children and adolescents below 18 years old.
Other medicines and Pergoveris
Tell your doctor or pharmacist if you are using, have recently used or might use any other medicines.
Do not use Pergoveris with other medicines in the same injection. You can use Pergoveris with a
licensed follitropin alfa preparation as separate injections, if prescribed by your doctor.
Pregnancy and breast-feeding
Do not use Pergoveris if you are pregnant or breast-feeding.
Driving and using machines
It is not expected that this medicine will affect your ability to drive or use machines.
Pergoveris contains sodium
Pergoveris contains less than 1 mmol sodium (23 mg) per dose, i.e. essentially “sodium-free”.
How to store Pergoveris
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label and the carton after EXP.
The expiry date refers to the last day of that month.
Store in a refrigerator (2°C-8°C). Do not freeze.
Store in the original package in order to protect from light.
Once opened, the pre-filled pen may be stored for a maximum of 28 days outside of the refrigerator (at 25°C).
Do not use Pergoveris if you notice any visible signs of deterioration, if the liquid contains particles or
is not clear.
After the injection, dispose of the used needle safely.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help to protect the environment.
Side Effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Most serious side effects
Contact your doctor straight away if you notice any of the below listed side effects. The doctor might ask you to stop using Pergoveris.
Allergic reactions
Allergic reactions such as rash, red skin, hives, swelling of your face with difficulty breathing can sometimes be serious. This side effect is very rare.
Ovarian hyperstimulation syndrome (OHSS)
• Lower abdominal pain together with nausea or vomiting. These may be the symptoms of ovarian hyperstimulation syndrome (OHSS). Your ovaries may have over-reacted to the
treatment and formed large sacs of fluid or cysts (see in section 2. under “Ovarian hyperstimulation syndrome (OHSS)”). This side effect is common. If this happens, your doctor will need to examine you as soon as possible.
• The OHSS may become severe with clearly enlarged ovaries, decreased urine production, weight gain, difficulty in breathing and/or possible fluid accumulation in your stomach or chest.
This side effect is uncommon (may affect up to 1 in 100 people).
• Complications of OHSS such as twisting of ovaries or blood clotting occur rarely (may affect up to 1 in 1,000 people).
• Serious blood clotting problems (thromboembolic events) usually with severe OHSS are found very rarely. This could cause chest pain, breathlessness, stroke or heart attack. In rare cases this
can also happen independently of OHSS (see in section 2. under “Blood clotting problems (thromboembolic events)”).
Other side effects
Very common (may affect more than 1 in 10 people):
• sacs of fluid within the ovaries (ovarian cysts)
• headache
• local reactions at the injection site such as pain, itching, bruising, swelling or irritation.
Common (may affect up to 1 in 10 people):
• diarrhoea
• breast pain
• feeling sick or vomiting
• abdominal or pelvic pain
• abdominal cramp or bloating.
Very rare (may affect up to 1 in 10,000 people):
• Your asthma may get worse.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side
effects not listed in this leaflet. You can also report side effects directly via
United Kingdom
Yellow Card Scheme
Website: www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or
Apple App Store
Ingredient
What Pergoveris contains
The active substances are follitropin alfa and lutropin alfa.
• Each pre-filled pen of Pergoveris (900 IU + 450 IU)/1.44 mL contains 900 IU (International Units) of follitropin alfa and 450 IU of lutropin alfa in 1.44 mL and can deliver six doses of Pergoveris 150 IU/75 IU.
The other ingredients are
• Sucrose, arginine monohydrochloride, poloxamer 188, methionine, phenol, disodium phosphate
dihydrate, sodium dihydrogen phosphate monohydrate and water for injections. Tiny amounts of concentrated phosphoric acid and sodium hydroxide are added to keep acidity levels (pH
levels) normal.
What Pergoveris looks like and contents of the pack
Pergoveris is presented as a clear, colourless to slightly yellow solution for injection in a mulitidose pre-filled pen:
• Pergoveris (900 IU + 450 IU)/1.44 mL is supplied in packs of 1 multidose pre-filled pen and 14 disposable injection needles.
Questions and answers of the customers
There are no questions yet, be the first to ask something for this product.
You Might Also Like
Original price was: £24.50.£16.50Current price is: £16.50.
- Keeps medicine at 15-25°C for 8-10hrs
- Stores 2 pens and diabetic kit
- Cold pack included
- Insulated, protective EVA case
- Ideal daily travel size
- 12.7 x 7.5 x 21.5cm
Original price was: £24.50.£16.50Current price is: £16.50.
- Availability: out of stock
Original price was: £89.50.£76.50Current price is: £76.50.
- KEEPS MEDICINE AT 2-8°C
- LASTS UP TO 36 HOURS
- LARGE INTERNAL SPACE
- x2 COLD PACKS INCLUDED
Original price was: £89.50.£76.50Current price is: £76.50.
- Availability: in stock
Original price was: £57.00.£49.50Current price is: £49.50.
- Keeps medicine between 2 – 8°C
- Stays cold for 12-18 hours*
- Ideal for travelling
- Water resistant
- Includes two gel packs
Original price was: £57.00.£49.50Current price is: £49.50.
- Availability: in stock
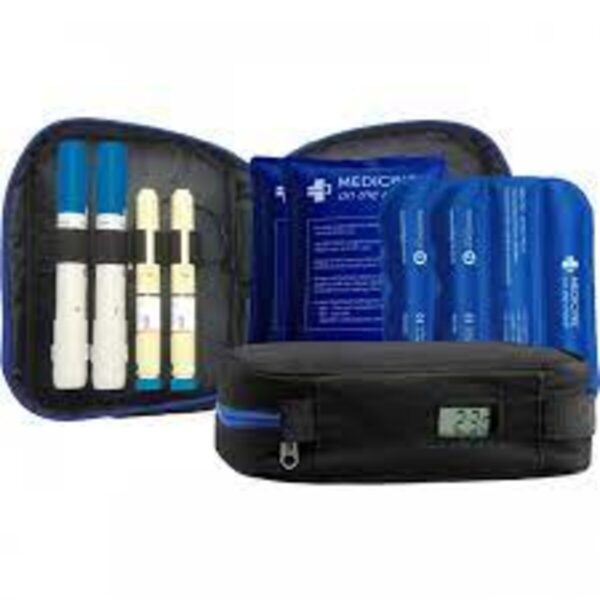
Original price was: £53.50.£49.50Current price is: £49.50.
- Keeps medicine between 2-8°C
- Stays cool for 12-18hrs
- Perfect for long haul flights
- Carries 4 pens/syringes
- Integrated digital thermometer
- Includes x2 cold packs and 2 buffer pads
- High quality canvas material
Original price was: £53.50.£49.50Current price is: £49.50.
- Availability: in stock
Other Products From This Seller
- Validated for 2-8°C for 30 to 36 hrs*
- Perfect for long-haul flights
- Carries pens, bottles, vials and syringes
- Manufactured from excellent quality materials
£102.50
- Availability: in stock
- Validated for 2-8°C for 18 to 24hrs*
- Perfect for long haul flights
- Carries pens, bottles, vials and syringes
- Integrated digital thermometer
- Manufactured from excellent quality materials
£69.45
- Availability: in stock
- Validated for 2-8°C for 15 to 20hrs*
- Perfect for long haul flights
- Carries pens, bottles, vials and syringes
- Integrated digital thermometer
- Manufactured from excellent quality materials
£54.50
- Availability: in stock
Steglatro Tablets – Ertugliflozin Tablets available in 2 strengths:
- Steglatro 5mg Tablets – Ertugliflozin 5mg Tablets
- Steglatro 15mg Tablets – Ertugliflozin 15mg Tablets
£49.50
- Availability: in stock
Original price was: £43.20.£39.50Current price is: £39.50.
Ursodeoxycholic Acid Tablets 250mg – Cholurso Tablets, 60 Tablets Introducing Cholurso Tablets the brand name of Ursodeoxycholic Acid Tablets 250mg – Your Solution for Gallstone Management and Liver Health Ursodeoxycholic Acid 250mg Tablets is a cutting-edge pharmaceutical formulation designed to positively influence bile composition, making it a versatile solution for various health concerns. Ursodeoxycholic Acid, […]
Learn MoreOriginal price was: £43.20.£39.50Current price is: £39.50.
- Availability: in stock
£156.00 – £210.00
Please note you need insulin pen needles to use Mounjaro Injection
Mounjaro Injection – Tirzepatide Injection, 4 Pre-filled Pens
Available in 6 strengths:
- Mounjaro 2.5mg Injection
- Mounjaro 5mg Injection
- Mounjaro 7.5mg Injection
- Mounjaro 10mg Injection
- Mounjaro 12.5mg Injection
- Mounjaro 15mg Injection
Learn More
£156.00 – £210.00
- Availability: in stock
Timoptol LA Gel – Timolol Eye Gel is available in 2 strengths
- Timoptol LA 0.5% Gel
- Timoptol LA 0.25% Gel
£7.50
- Availability: in stock
Original price was: £43.50.£39.75Current price is: £39.75.
One-Alpha Drops 2mcg/1ml – Alfacalcidol Drops 2mcg/1ml
Learn MoreOriginal price was: £43.50.£39.75Current price is: £39.75.
- Availability: in stock
Original price was: £5.99.£5.39Current price is: £5.39.
Ideal for irrigating and cleansing the eye, or a wound
Learn MoreOriginal price was: £5.99.£5.39Current price is: £5.39.
- Availability: in stock
Ultimate Surefit Erection Ring Set 15222 – Constrictor Rings For Erectile Dysfunction Experience Confidence and Pleasure with the Ultimate Surefit maintenance ring set – Your Key to Intimate Well-being Introducing the Ultimate Surefit Erection Ring, a premium solution meticulously designed for those seeking to overcome the challenges of erectile dysfunction. Crafted with precision and comfort […]
Learn More£58.50
- Availability: in stock