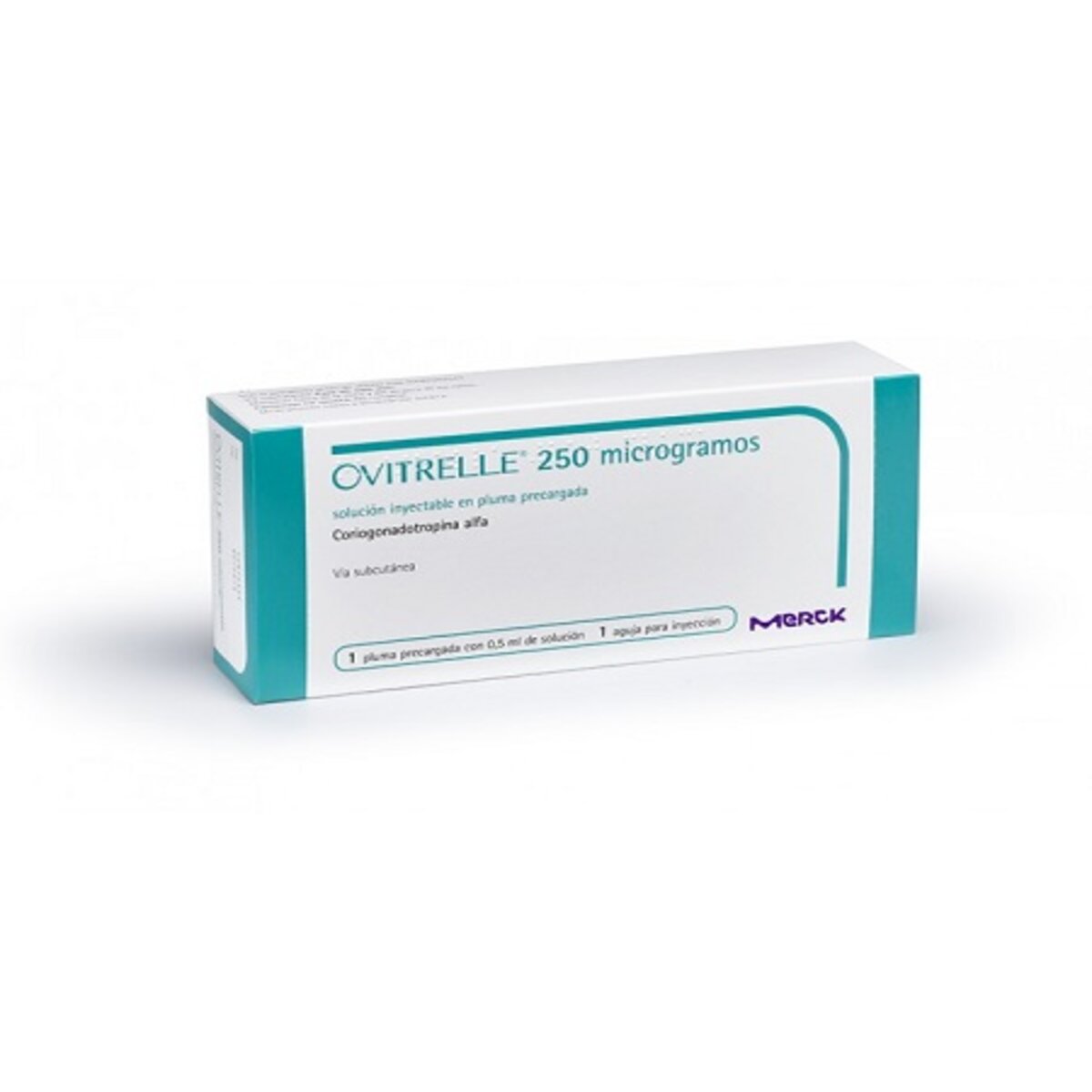
Ovitrelle Injection Pre-filled Syringe, 1 Pre-filled Syringe
Fertility treatment in women
Active Ingredient: choriogonadotropin alfa
Fertility treatment in women
Active Ingredient: choriogonadotropin alfa
£56.49
CompareCompare- Description
- Additional Information
- Brand
- How To Use
- Product Details
- Side Effects
- Ingredients
- How to Store
- Patient Information leaflet
- Reviews (0)
- Questions & Answers
Ovitrelle Injection Pre-filled Syringe, 1 Pre-filled Syringe
Ovitrelle Injection Pre-filled Syringe is a revolutionary new injectable fertility medication that can help you conceive quickly and easily. Ovitrelle contains choriogonadotropin alfa, a synthetic hormone that is similar to the naturally-occurring chorionic gonadotropin hormone. This hormone is essential for reproduction and fertility, and by using Ovitrelle, you can ensure that you are getting the optimal amount of this important substance. In addition, Ovitrelle is easy to use, with a pre-filled Syringe that makes administration simple and quick. If you are struggling to conceive, or if you have been unsuccessful with other fertility treatments, Ovitrelle may be the answer you have been looking for.
What Ovitrelle is used for?
Ovitrelle is used together with other medicines:
- To help develop and ripen several follicles (each containing an egg) in women undergoing assisted reproductive technologies (procedure that may help you to become pregnant) such as ‘in vitro fertilisation’. Other medicines will be given first to bring about the growth of several follicles.
- To help release an egg from the ovary (ovulation induction) in women who cannot produce eggs (‘anovulation’), or women who produce too few eggs (‘oligo-ovulation’). Other medicines will be given first to develop and ripen the follicles.
Ovitrelle Injection Pre-filled Syringe Reviews
After using Ovitrelle Injection Pre-filled Syringe, it’s helpful to let others know about your experience. Reviews of an item help other users know that medicines received have helped the condition it is claimed for, how well the treatment worked or any issues to be aware of. We invite our users to leave a review of both their treatment and of the service provided. Click on the reviews tab to see if there has been feedback on this item.
What is the price of Ovitrelle Injection Pre-filled Syringe?
The price of Ovitrelle Injection Pre-filled Syringe is £56.49
Where can you buy Ovitrelle Injection Pre-filled Syringe?
You can buy Ovitrelle Injection Pre-filled Syringe at Dock Pharmacy Essex UK, UK Online Pharmacy.
Can you buy Ovitrelle Injection Pre-filled Syringe Over the counter?
Ovitrelle Injection Pre-filled Syringe is not available to buy over the counter. You need a prescription to buy Ovitrelle Injection Pre-filled Syringe
| Brand | |
|---|---|
Brand
OVITRELLE
How To Use
How To Use Ovitrelle Injection Pre-filled Syringe
Always use this medicine exactly as your doctor has told you.
Check with your doctor or pharmacist if you are not sure. How much to use
- The recommended dose is 1 pre-filled syringe (250 micrograms/0.5 mL) given as a single injection. • Your doctor will have explained exactly when to give the injection. Using this medicine
- Ovitrelle is intended for subcutaneous use that means given by injection under the skin.
- Each pre-filled syringe is for single use only. Only clear solution without particles should be used.
- Your doctor or nurse will show you how to use the Ovitrelle pre-filled syringe to inject the medicine.
- Inject Ovitrelle as your doctor or nurse taught you.
- After the injection, dispose of the used syringe safely.
If you administer Ovitrelle to yourself, please carefully read the following instructions:
- Wash your hands. It is important that your hands and the items you use are as clean as possible. 2. Assemble everything you need. Please note that alcohol swabs are not contained in the package. Find a clean area and lay out everything: – two alcohol swabs, – one pre-filled syringe containing the medicinal product.
- Injection: Immediately inject the solution: Your doctor or nurse will have already advised you where to inject (e.g. tummy, front of thigh). Wipe the chosen area with an alcohol swab.
Firmly pinch the skin together and insert the needle for injection at a 45° to 90° angle using a dart-like motion. Inject under the skin, as you were taught.
Do not inject directly into a vein.
Inject the solution by pushing gently on the plunger. Take as much time as you need to inject all the solution.
Immediately withdraw the needle and clean the skin with an alcohol swab using a circular motion. 4. Dispose of all used items: Once you have finished your injection, immediately discard the empty syringe in a sharps container.
Any unused solution must be discarded.
If you use more Ovitrelle than you should The effects of an overdose of Ovitrelle are unknown, nevertheless there is a possibility that ovarian hyperstimulation syndrome (OHSS) may occur, which is further described in section 4.
If you forget to use Ovitrelle
If you forget to use Ovitrelle, please talk to your doctor as soon as you notice. If you have any further questions on the use of this medicine, ask your doctor or pharmacist
Product Details
Product Information of Ovitrelle Injection Pre-filled Syringe
What do you need to know before you use Ovitrelle
Do not use Ovitrelle · if you are allergic to choriogonadotropin alfa or any of the other ingredients of this medicine (listed in section 6). · if you have a tumour in your hypothalamus or pituitary gland (both are parts of the brain). · if you have large ovaries or sacs of fluid within the ovaries (ovarian cysts) of unknown origin. · if you have unexplained vaginal bleeding. · if you have cancer of your ovaries, womb or breast. · if you have severe inflammation of your veins or blood clotting in your veins (active thromboembolic disorders). · if you have any condition that usually makes a normal pregnancy impossible, such as menopause or early menopause (ovarian failure), or malformations of sexual organs. Do not use Ovitrelle if any of the above applies to you. If you are not sure, talk to your doctor before using this medicine. Warnings and precautions Before the treatment is started, your and your partner’s fertility should be evaluated by a doctor experienced in the treatment of fertility problems. Ovarian hyperstimulation syndrome (OHSS) This medicine may increase your risk of developing OHSS. This is when your follicles develop too much and become large cysts. If you get lower abdominal pain, gain any weight rapidly, feel sick or are vomiting, or have difficulty in breathing, do not give yourself the Ovitrelle injection and talk to your doctor straight away (see section 4). If you are developing OHSS, you may be told not to have sex or to use a barrier contraceptive method for at least four days. The risk of OHSS is reduced if the usual dose of Ovitrelle is used, and if you are monitored closely throughout your treatment cycle (e.g. blood tests for estradiol levels and ultrasound). Multiple pregnancy and/or birth defects When using Ovitrelle, you have a higher risk of being pregnant with more than one child at the same time (‘multiple pregnancy’, usually twins) than if you conceived naturally. Multiple pregnancy may lead to medical complications for you and your babies. When undergoing assisted reproductive technologies, the risk of having a multiple pregnancy is related to your age, the quality and the number of fertilised eggs or embryos placed inside you. Multiple pregnancies and specific characteristics of couples with fertility problems (e.g. age) may also be associated with an increased chance of birth defects. The risk of multiple pregnancy is reduced if you are monitored closely throughout your treatment cycle (e.g. blood tests for estradiol levels and ultrasound). Ectopic pregnancy Pregnancy outside of the womb (an ectopic pregnancy) may occur in women with damaged fallopian tubes (the tubes which carry the egg from the ovary to the womb). Therefore, your doctor should perform an early ultrasound examination to rule out the possibility of pregnancy outside the womb. Miscarriage When undergoing assisted reproductive technologies or stimulation of your ovaries to produce eggs, you are more likely to have a miscarriage than the average woman. Blood clotting problems (thromboembolic events) Talk to your doctor before using Ovitrelle if you or a member of your family have ever had blood clots in the leg or in the lung, or a heart attack or stroke. You may be at a higher risk of serious blood clots or existing clots might become worse with Ovitrelle treatment. Tumours of sexual organs There have been reports of tumours in the ovaries and other sex organs, both benign and malignant, in women who have undergone multiple regimens for infertility treatment. Pregnancy tests If you do a pregnancy test with serum or urine after use of Ovitrelle, and up to ten days later, it may happen that you get a false positive test result. If you are not sure, talk to your doctor. Children and adolescents Ovitrelle is not for use in children and adolescents. Other medicines and Ovitrelle Tell your doctor if you are taking, have recently taken or might take any other medicines. Pregnancy and breast-feeding Do not use Ovitrelle if you are pregnant or breast-feeding. If you are pregnant or breast-feeding, ask your doctor for advice before taking this medicine. Driving and using machines It is not expected that Ovitrelle will affect your ability to drive and use machines. Ovitrelle contains sodium This medicine contains less than 1 mmol sodium (23 mg) per dose, that is to say essentially “sodiumfree”.
Side Effects
Side Effects of Ovitrelle Injection Pre-filled Syringe
Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. Stop using Ovitrelle and see a doctor straight away if you notice any of the following serious side effects – you may need urgent medical treatment: · Allergic reactions such as rash, fast or uneven pulse, swelling of your tongue and throat, sneezing, wheezing, or serious breathing difficulty are very rare (may affect up to 1 in 10,000 people). · Lower abdominal pain, abdominal distension or abdominal discomfort together with nausea (feeling sick) or vomiting may be the symptoms of ovarian hyperstimulation syndrome (OHSS). This may indicate that the ovaries over-reacted to the treatment and that large ovarian cysts developed (see also in section 2. under “Ovarian hyperstimulation syndrome”). This event is common (may affect up to 1 in 10 people). · The OHSS may become severe with clearly enlarged ovaries, decreased urine production, weight gain, difficulty in breathing and possible fluid accumulation in your stomach or chest. This event is uncommon (may affect up to 1 in 100 people). · Serious blood clotting complications (thromboembolic events) sometimes independent of OHSS may be found very rarely. This could cause chest pain, breathlessness, stroke or heart attack (see also in section 2. under “Blood clotting problems”). Other side effects Common (may affect up to 1 in 10 people) · Headache. · Local reactions at the injection site, such as pain, redness or swelling. Uncommon (may affect up to 1 in 100 people) · Diarrhoea. Reporting of side effects If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via United Kingdom Yellow Card Scheme Website: www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store By reporting side effects you can help provide more information on the safety of this medicine.
Ingredients
Ingredients of Ovitrelle Injection Pre-filled Syringe
What Ovitrelle contains
- The active substance is choriogonadotropin alfa, produced by recombinant DNA technology.
- Each pre-filled pen contains 250 micrograms choriogonadotropin alfa in 0.5 mL (equivalent to approximately 6,500 International Units, IU).
- The other ingredients are mannitol, methionine disodium phosphate dihydrate, sodium dihydrogen phosphate monohydrate, poloxamer 188, phosphoric acid (for pH adjustment), sodium hydroxide (for pH adjustment) and water for injections.
What Ovitrelle looks like and contents of the pack
- Ovitrelle comes as a clear, colourless to slightly yellow liquid for injection in a pre-filled pen.
- Each pen contains 0.5 mL of solution.
- It is supplied in packs of 1 pre-filled pen and 2 injection needles (one spare).
How to Store
How to Store Ovitrelle Injection Pre-filled Syringe
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label and carton after EXP. The expiry date refers to the last day of that month. Store in a refrigerator (2°C-8°C). Do not freeze. Do not use Ovitrelle if you notice any signs of deterioration, if the liquid contains particles or is not clear. Do not throw away any medicine via wastewater or household waste. Ask your pharmacist how to throw away medicines no longer used. These measures will help to protect the environment.
Patient Information leaflet
Click here for the Patient Information leaflet of Ovitrelle Injection Pre-filled Syringe
Please read before using the product
Only logged in customers who have purchased this product may leave a review.
Questions and answers of the customers
There are no questions yet, be the first to ask something for this product.
You Might Also Like
Fertility treatment in women
Active Ingredient: choriogonadotropin alfa
Learn More
£56.49
- Availability: in stock
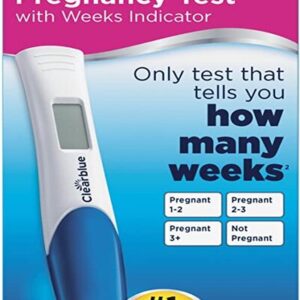
Clearblue Digital Pregnancy Test with Conception Indicator is accurate as a doctor’s urine test* over 99% accurate at telling you if you’re pregnant, from the day your period is due.
Learn More£9.99
- Availability: in stock
Original price was: £560.30.£515.50Current price is: £515.50.
- Treat infertility In women
- Treat Low sperm count in men
- Prescription Required.
Original price was: £560.30.£515.50Current price is: £515.50.
- Availability: in stock
Other Products From This Seller
- Validated for 2-8°C for 30 to 36 hrs*
- Perfect for long-haul flights
- Carries pens, bottles, vials and syringes
- Manufactured from excellent quality materials
£102.50
- Availability: in stock
- Validated for 2-8°C for 18 to 24hrs*
- Perfect for long haul flights
- Carries pens, bottles, vials and syringes
- Integrated digital thermometer
- Manufactured from excellent quality materials
£69.45
- Availability: in stock
- Validated for 2-8°C for 15 to 20hrs*
- Perfect for long haul flights
- Carries pens, bottles, vials and syringes
- Integrated digital thermometer
- Manufactured from excellent quality materials
£54.50
- Availability: in stock
Steglatro Tablets – Ertugliflozin Tablets available in 2 strengths:
- Steglatro 5mg Tablets – Ertugliflozin 5mg Tablets
- Steglatro 15mg Tablets – Ertugliflozin 15mg Tablets
£49.50
- Availability: in stock
Original price was: £43.20.£39.50Current price is: £39.50.
Ursodeoxycholic Acid Tablets 250mg – Cholurso Tablets, 60 Tablets Introducing Cholurso Tablets the brand name of Ursodeoxycholic Acid Tablets 250mg – Your Solution for Gallstone Management and Liver Health Ursodeoxycholic Acid 250mg Tablets is a cutting-edge pharmaceutical formulation designed to positively influence bile composition, making it a versatile solution for various health concerns. Ursodeoxycholic Acid, […]
Learn MoreOriginal price was: £43.20.£39.50Current price is: £39.50.
- Availability: in stock
£156.00 – £210.00
Please note you need insulin pen needles to use Mounjaro Injection
Mounjaro Injection – Tirzepatide Injection, 4 Pre-filled Pens
Available in 6 strengths:
- Mounjaro 2.5mg Injection
- Mounjaro 5mg Injection
- Mounjaro 7.5mg Injection
- Mounjaro 10mg Injection
- Mounjaro 12.5mg Injection
- Mounjaro 15mg Injection
Learn More
£156.00 – £210.00
- Availability: in stock
Timoptol LA Gel – Timolol Eye Gel is available in 2 strengths
- Timoptol LA 0.5% Gel
- Timoptol LA 0.25% Gel
£7.50
- Availability: in stock
Original price was: £43.50.£39.75Current price is: £39.75.
One-Alpha Drops 2mcg/1ml – Alfacalcidol Drops 2mcg/1ml
Learn MoreOriginal price was: £43.50.£39.75Current price is: £39.75.
- Availability: in stock
Original price was: £5.99.£5.39Current price is: £5.39.
Ideal for irrigating and cleansing the eye, or a wound
Learn MoreOriginal price was: £5.99.£5.39Current price is: £5.39.
- Availability: in stock
Ultimate Surefit Erection Ring Set 15222 – Constrictor Rings For Erectile Dysfunction Experience Confidence and Pleasure with the Ultimate Surefit maintenance ring set – Your Key to Intimate Well-being Introducing the Ultimate Surefit Erection Ring, a premium solution meticulously designed for those seeking to overcome the challenges of erectile dysfunction. Crafted with precision and comfort […]
Learn More£58.50
- Availability: in stock
















Reviews
There are no reviews yet.